Abstract
Objectives
To evaluate short-term clinical efficacy, complications and possible passive stent expansion of transjugular intrahepatic portosystemic shunt (TIPS) creation using the new controlled expansion ePTFE covered stent (VCX), for portal hypertension complications.
Methods
Between 7/2016 and 3/2018, 75 patients received TIPS using VCX. Thirty-nine patients with VCX dilated with an 8-mm angioplasty balloon underwent computed tomography (CT) study during follow-up and CT data were used to measure stent diameter. The CT measurement technique was validated by ex vivo experiment.
Results
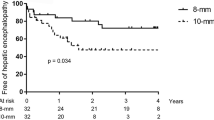
TIPS indications were: refractory ascites (n = 45), variceal bleeding (n = 22), other (n = 8). Mean follow-up was 5.8 months (± 4.5, range 1–20). In 69 patients, TIPS was dilated to 8 mm of diameter reaching the hemodynamic target of a portosystemic pressure gradient (PSG) < 12 mmHg. In six patients, not reaching the hemodynamic target the stent was dilated to 10 mm of diameter during the same session with a final PSG < 12 mmHg. Overall clinical success was achieved in 66/75 (88%) patients (80% in refractory ascites, 95% variceal bleeding, 100% other). Grade II–III encephalopathy was observed in five patients (6%). TIPS revision with stent dilatation to 10 mm was performed in seven patients: in three patients with ascites persistence, without evidence of stent dysfunction and in four patients for stent stenosis. One patient underwent stent reduction. Fourteen patients (18%) died during follow-up of causes not related to TIPS. Five patients (6%) underwent liver transplant. No passive stent expansion was detected by CT measurements.
Conclusion
VCX for TIPS creation retains its diameter over a short-term period and is associated with a good clinical outcome with a reasonably low complication rate.






Similar content being viewed by others
References
Rossle M. TIPS: 25 years later. J Hepatol. 2013;59:1081–93.
Riggio O, Angeloni S, Salvatori FM, et al. Incidence, natural history, and risk factors of hepatic encephalopathy after transjugular intrahepatic portosystemic shunt with polytetrafluoroethylene-covered stent grafts. Am J Gastroenterol. 2008;103(11):2738–46.
Riggio O, Nardelli S, Moscucci F, et al. Hepatic encephalopathy after transjugular intrahepatic portosystemic shunt. Clin Liv Dis. 2012;16(1):133–46.
Casadaban LC, Parvinian A, et al. Clearing the confusion over hepatic encephalopathy after TIPS creation: incidence, prognostic factors, and clinical outcomes. Dig Dis Sci. 2015;60(4):1059–66.
Bureau C, Garcia-Pagan JC, Otal P, et al. Improved clinical outcome using polytetrafluoroethylene-coated stents for TIPS: results of a randomized study. Gastroenterology. 2004;126:469–75.
Bureau C, Pagan JC, Layrargues GP, et al. Patency of stents covered with polytetrafluoroethylene in patients treated by transjugular intrahepatic portosystemic shunts: long-term results of a randomized multicentre study. Liv Int. 2007;27:742–7.
Sarfeh IJ, Rypins EB. Partial versus total portacaval shunt in alcoholic cirrhosis. Results of a prospective, randomized clinical trial. Ann Surg. 1994;219:353–61.
Pieper CC, Sprinkart AM, Nadal J, et al. Postinterventional passive expansion of partially dilated transjugular intrahepatic portosystemic shunt stents. J Vasc Interv Radiol. 2015;26(3):388–94.
Gaba RC, Parvinian A, Minocha J, et al. Should transjugular intrahepatic portosystemic shunt stent grafts be underdilated? J Vasc Interv Radiol. 2015;26(3):382–7.
Borghol S, Perarnau JM, Pucheux J, et al. Short- and long-term evolution of the endoluminal diameter of underdilated stents in transjugular intrahepatic portosystemic shunt. Diagn Interv Imaging. 2016;97(11):1103–7.
Pieper CC, Jansen C, Meyer C, et al. Prospective evaluation of passive expansion of partially dilated transjugular intrahepatic portosystemic shunt stent grafts—a three-dimensional sonography study. J Vasc Interv Radiol. 2017;28(1):117–25.
Mollaiyan A, Bettinger D, Rössle M. The underdilation of nitinol stents at TIPS implantation: solution or illusion? Eur J Radiol. 2017;89:123–8.
Praktiknjo M, Lehmann J, Fischer S, et al. Novel diameter controlled expansion TIPS (Viatorr CX®) graft reduces readmission compared to regular covered TIPS graft and bare metal graft. J Hepatol. 2017;66:S33–62.
de Franchis R, Baveno V. Faculty. Revising consensus in portal hypertension: report of the Baveno V consensus workshop on methodology of diagnosis and therapy in portal hypertension. J Hepatol. 2010;53(4):762–8.
Miraglia R, Maruzzelli L, Cortis K, et al. Radiation exposure in transjugular intrahepatic portosystemic shunt creation. Cardiovasc Intervent Radiol. 2016;39:210–7.
Miraglia R, Gerasia R, Maruzzelli L, et al. Radiation doses to operators performing transjugular intrahepatic portosystemic shunt using a flat-panel detector-based system and ultrasound guidance for portal vein targeting. Eur Radiol. 2017;27(5):1783–6.
La Mura V, Abraldes JG, Berzigotti A, et al. Right atrial pressure is not adequate to calculate portal pressure gradient in cirrhosis: a clinical–hemodynamic correlation study. Hepatology. 2010;51:2108–16.
Bauer J, Johnson S, Durham J, et al. The role of TIPS for portal vein patency in liver transplant patients with portal vein thrombosis. Liv Transpl. 2006;12(10):1544–51.
Riggio O, Ridola L, Angeloni S, et al. Clinical efficacy of transjugular intrahepatic portosystemic shunt created with covered stents with different diameters: results of a randomized controlled trial. J Hepatol. 2010;53:267–72.
Miraglia R, Maruzzelli L, Tuzzolino F, et al. Transjugular intrahepatic portosystemic shunts in patients with cirrhosis with refractory ascites: comparison of clinical outcomes by using 8- and 10-mm PTFE-covered stents. Radiology. 2017;284(1):281–8.
Sauerbruch T, Mengel M, Dollinger M, et al. Prevention of rebleeding from esophageal varices in patients with cirrhosis receiving small-diameter stents versus hemodynamically controlled medical therapy. Gastroenterology. 2015;149:660–88.e1.
Wang Q, Lv Y, Bai M, et al. Eight millimetre covered TIPS does not compromise shunt function but reduces hepatic encephalopathy in preventing variceal rebleeding. J Hepatol. 2017;67(3):508–16.
Schepis F, Vizzutti F, Garcia-Tsao G, et al. Under-dilated TIPS associate with efficacy and reduced encephalopathy in a prospective, non-randomized study of patients with cirrhosis. Clin Gastroenterol Hepatol. 2018;16(7):1153–62.e7.
Fagiuoli S, Bruno R, Debernardi Venon W, et al. Consensus conference on TIPS management: techniques, indications, contraindications. Dig Liv Dis. 2017;49(2):121–37.
García-Pagán JC, Caca K, Bureau C, et al. Early use of TIPS in patients with cirrhosis and variceal bleeding. N Engl J Med. 2010;25:2370–9.
Bureau C, Thabut D, Oberti F, et al. Transjugular intrahepatic portosystemic shunts with covered stents increase transplant-free survival of patients with cirrhosis and recurrent ascites. Gastroenterology. 2017;152(1):157–63.
Farsad K, Kolbeck KJ, Keller FS, et al. Primary creation of an externally constrained TIPS: a technique to control reduction of the portosystemic gradient. AJR Am J Roentgenol. 2015;04:868–71.
Cui J, Smolinski SE, Liu F, et al. Incrementally expandable transjugular intrahepatic portosystemic shunts: single-center experience. AJR Am J Roentgenol. 2018;210(2):438–46.
Rabei R, Mathesovian S, Tasse J, et al. Primary constrained TIPS for treating refractory ascites or variceal bleeding secondary to hepatic cirrhosis. Br J Radiol. 1083;2018(91):20170409.
https://www.goremedical.com/products/viatorr—controlled-expansion. Accessed 28 Jun 2018.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for Publication
For this type of study consent for publication is not required.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Informed Consent
This study has obtained IRB approval from IRCCS-ISMETT and the need for informed consent was waived.
Rights and permissions
About this article
Cite this article
Miraglia, R., Maruzzelli, L., Di Piazza, A. et al. Transjugular Intrahepatic Portosystemic Shunt Using the New Gore Viatorr Controlled Expansion Endoprosthesis: Prospective, Single-Center, Preliminary Experience. Cardiovasc Intervent Radiol 42, 78–86 (2019). https://doi.org/10.1007/s00270-018-2040-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-018-2040-y