Abstract
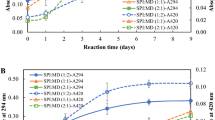
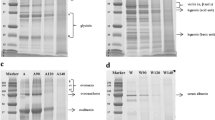
This study aimed to evaluate the effects of pH-shifting treatment combined with microbial transglutaminase (MTG)-mediated modification on the structure, digestibility, and IgE-binding of glycinin. Glycinin was incubated in acidic (pH 1.0) or alkaline (pH 13.0) solutions to induce protein structure to unfolding followed by refolding for 1 h at pH 7.0. Afterwards, glycinin was incubated with MTG under appropriate conditions. Sodium dodecyl sulfate polyacrylamide gel electropheresis(SDS-PAGE), circular dichroism, UV absorption spectra, and surface hydrophobicity were considered to measure the changes in the structure of glycinin. The digestibility and IgE-binding of glycinin were determined by Tricine-SDS-PAGE and ELISA, respectively. The results showed that pH 1.0 shifting caused the unfolding of the spatial structure of glycinin and the formation of some polymers via disulfide bond. After glycinin was incubated with MTG, the protein preferentially underwent embedding and folding. The acidic compound-modified glycinin was stable for digestion. Under pH 13.0 shifting treatment, glycinin was partially hydrolyzed, and the MTG-modified alkaline-treated glycinin was slightly affected with a good digestibility. Compound modification could reduce the IgE-binding of glycinin, especially under alkaline conditions. Our findings suggested that alkaline pH shifting combined with MTG cross-linking can be an efficient approach to reduce the IgE-binding of glycinin with a labile digestion.





Similar content being viewed by others
References
E.Y. Kim, K.B. Hong, H.J. Suh, H.S. Choi, Food Funct. 6(11), 3512–3521 (2015)
S. Hu, H. Liu, S. Qiao, P. He, X. Ma, W. Lu, J Agric Food Chem 61(18), 4406–4410 (2013)
B. Liu, D. Teng, Y.L. Yang, X.M. Wang, J.H. Wang, Process Biochem. 47(2), 280–287 (2012)
E. Penas, G. Prestamo, F. Polo, R. Gomez, Food Chem. 99(3), 569–573 (2006)
Y. Katz, P. Gutierrez-Castrellon, M.G. Gonzalez, R. Rivas, B.W. Lee, P. Alarcon, Clin Rev Allergy Immunol 46(3), 272–281 (2014)
H. Yang, A.S. Yang, J.Y. Gao, H.B. Chen, J. Food Sci. 79(11), C2157–C2163 (2014)
J. Chen, J. Wang, P. Song, X. Ma, Food Chem. 162, 27–33 (2014)
T. Wang, G.X. Qin, Z.W. Sun, Y. Zhao, Crit. Rev. Food Sci. Nutr. 54(7), 850–862 (2014)
K. Prak, B. Mikami, T. Itoh, T. Fukuda, N. Maruyama, S. Utsumi, Acta Crystallogr Sect F 69(8), 937–941 (2013)
B. Yuan, J. Ren, M. Zhao, D. Luo, L. Gu, LWT Food Sci Technol 46(2), 453–459 (2012)
L. Tuppo, R. Spadaccini, C. Alessandri, A. Mari, R. Boelens, Biopolymers 102(5), 416–425 (2014)
W. Chen, J.Y. Gao, H.B. Chen, Food Sci. 23, 391–394 (2010)
Z. Li, Y. Luo, M. Jiang, J. Aquat, Food Product Technol. 25(3), 350–357 (2015)
O.E. Makinen, E. Zannini, P. Koehler, E.K. Arendt, Food Chem. 196, 17–24 (2016)
D. Sung, K.M. Ahn, S.Y. Lim, S. Oh, J Sci Food Agric 94(12), 2482–2487 (2014)
X. Ma, D. Lozano, H. Ojalvo, R. Chen, E. Lopez Fandiño, I. Molina, Food Sci. Emerg. 29, 143–150 (2015)
L. Monaci, S.L. Bavaro, E. De Angelis, L. Monaci, Food Funct. (2017)
G.X. Qin, Z.W. Sun, Y. Zhao, Food Agric. Immunol. 21(3), 23–263 (2010)
Y. Li, L.Z, Jiang, Y. Yang, R. Wang, F.F. Han, Biomed. Res. Int. (2016)
J. Jiang, J. Chen, Y.L. Xiong, J Agric Food Chem 57(16), 7576–7583 (2009)
Y. Liang, H.G. Kristinsson, Food Res. Int. 40(6), 668–678 (2007)
G. Rabbani, E. Ahmad, N. Zaidi, S. Fatima, R.H. Khan, Cell Biochem. Biophys. 62(3), 487–499 (2012)
R. Roychaudhuri, G. Sarath, M. Zeece, J. Markwell, Biochem. Biophys. Acta 1699(1–2), 207–212 (2004)
Y.H. Chang, S.Y. Shiau, F.B. Chen, F.R. Lin, LWT Food Sci Technol 44(4), 1107–1112 (2011)
R. Porta, C.V.L. Giosafatto, P. di Pierro, A. Sorrentino, L. Mariniello, Amino Acids 44(1), 285–292 (2013)
C. Xia, L. Wang, Y. Dong, S. Zhang, S.Q. Shi, L. Cai, J. Li, RSC Adv. 5(101), 82765–82771 (2015)
E.F.E. Babiker, M.A.S. han, N. Matsudomi, A. Kato, Food Res. Int. 29(7), 627–634 (1996)
L.J. Kang, Y. Matsumura, K. Ikura, J Agric Food Chem 42(1), 159–165 (1994)
Y. Chanyongvorakul, Y. Matsumura, M. Nonaka, M. Motoki, T. Mori, J. Food Sci. 60(493), 483–488 (1995)
C.H. Tang, H. Wu, Z. Chen, Food Res. Int. 39(1), 87–97 (2006)
M.Y. Han, H.Z. Zu, X.L. Xu, G.H. Zhou, J Food Process Preserv 39(3), 309–317 (2015)
A.S. Yang, J.H. Xia, Y.Q. Gong, H.B. Chen, Modern Food Sci. Technol. 33(2), 55–60 (2017)
D.A. Clare, G. Gharst, S.J. Maleki, T.H. Sanders, J Agric Food Chem 56(22), 10913–10921 (2008)
Y. Zhou, J.S. Wang, X.J. Yang, D.H. Lin, Y.F. Gao, Y.J. Su, S. Yang, Y.J. Zhang, J.J. Zheng, Int. J. Food Sci. 2013, 1–8 (2013)
M.B. Villas-Boas, M.A. Fernandes, R. de Lima Zollner, F.M. Netto, Int. Dairy J. 25(2), 123–131 (2012)
E. Dekking, P. Van Veelen, A. De Ru, E. Kooy Winkelaar, T. Groneveld, W. Nieuwenhuizen, F. Koning, J. Cereal Sci. 47(2), 339–346 (2008)
D.H.J. Hou, S.K.C. Chang, J Agric Food Chem 52(12), 3792–3800 (2004)
G.L. Ellman, Arch. Biochem. Biophys. 82(1), 70–77 (1959)
P. Eyer, D. Kiderlen, F. Worek, Anal. Biochem. 312(2), 224–227 (2003)
N. Sreerama, R.W. Woody, Anal. Biochem. 287(2), 252–260 (2000)
M. Amigo Benavent, A. Clemente, P. Ferranti, S. Caira, M.D. del Castillo, Food Chem. 129(4), 1598–1605 (2011)
A.S. Yang, C.Y. Long, J.H. Xia, P. Tong, Y.F. Cheng, Y. Wang, H.B. Chen, J Sci Food Agric 97(1), 199–206 (2016)
J. Xiao, C. Shi, L. Zhang, Y. Li, J. Qi, Y. Wang, Q. Huang, Food Res. Int. 89(1), 540–548 (2016)
J. Jiang, Y.L. Xiong, M.C. Newman, G.K. Rentfrow, Food Chem. 132(4), 1944–1950 (2012)
C.Y. Zhao, H.F. Liu, M. Fu, X.H. Zhao, CyTA - J Food 14(1), 138–144 (2015)
A.L. Gaspar, S.P. de Goes Favoni, Food Chem. 171, 315–322 (2015)
P. Zhang, T. Hu, S. Feng, Q. Xu, T. Zheng, M. Zhou, X. Chu, X. Huang, X. Lu, S. Pan, E.C. Li-Chan, H. Hu, Ultrason. Sonochem. 29, 380–387 (2016)
X. Rui, Y. Fu, Q. Zhang, W. Li, F. Zare, X. Chen, M. Jiang, M. Dong, LWT Food Sci Technol 71, 234–242 (2016)
Acknowledgments
The authors are grateful for financial support of National Natural Science Foundation of China (No. 31460439, 31760453), National High Technology Research and Development Program of China (863 Program, No. 2013AA102205), Young Scientist Training Program of Jiangxi Province (No. 20122BCB23006), International Science & Technology Cooperation Program of Jiangxi Province (No. 20142BDH80002).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Fig. S1
Flow chart of the pH-shifting combined with MTG-mediated modification of glycinin (DOCX 2694 kb)
Rights and permissions
About this article
Cite this article
Yang, A., Bai, J., Xia, J. et al. Structure Changes in Relation to Digestibility and IgE-Binding of Glycinin Induced by pH-Shifting Combined with Microbial Transglutaminase-Mediated Modification. Food Biophysics 14, 269–277 (2019). https://doi.org/10.1007/s11483-019-09580-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11483-019-09580-4